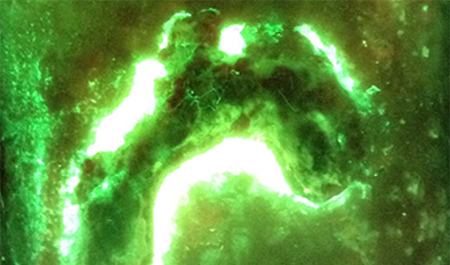
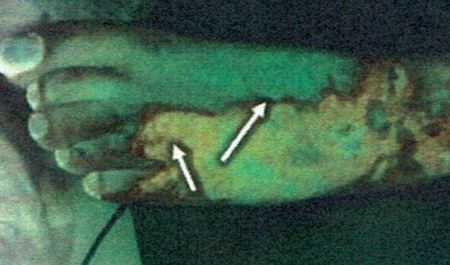
Paediatric burn injuries are vulnerable to severe complications, most often infection, making prompt and precise diagnosis of bacterial bioburden vital to preventing detrimental consequences and optimizing patients’ outcomes. Currently, burn wounds are assessed for infection via examining the clinical signs and symptoms of infection, which can be confirmed by swab culture analysis. While the former approach is subjective and experience-dependant, the latter technique is susceptible to missing subsurface, biofilm-associated colonization and any peripheral bacterial burden, and also delays confirmation by up to 5 days. The MolecuLight i:X is a handheld, non-contact fluorescence imaging device, which can reveal real-time information about clinically-significant levels of bacteria and their biodistribution in surface and subsurface burn wound tissues. We conducted a single-centre observational study to assess the device efficacy in identifying critical bacterial levels in paediatric burn wounds and to test the children’s compliance and the overall feasibility of the device integration into the current diagnostic practice. Ten patients with sixteen wounds were recruited and assessed for the presence or absence of clinical signs and symptoms of infection and the presence or absence of bacterial fluorescence on images, with swabs taken to confirm findings. Results demonstrate the device’s ability to visualize clinically-significant bacterial burden and to localize distribution of pathogens. All clinicians agreed on the high compliance with the device and high feasibility of incorporating the device into routine wound assessments. The results of this study may pave the way towards including bacterial fluorescence imaging into the standard diagnostic algorithm for paediatric burn population.
Back to All Clinical Evidence
Burns, Acute Wounds
Utility of MolecuLight i:X for Managing Bacterial Burden in Paediatric Burns
88% of wounds where bacterial fluorescence was detected in peripheries (in regions not typically swabbed)
100% of pediatric patients were compliant with the MolecuLight imaging procedure
93% of clinicians who reported highest practicality of incorporating MolecuLight i:X into routine diagnostic practice
References
Journal of Burn Care & Research, 21 September 2019